|
11/20/2022 0 Comments How long is half life Energy of levels in MeV.įor adjustment and calibration of equipment, it is convenient to have nuclides emitting either only alpha or only beta particles. Energy diagrams of decays: (A) β +-decay of 22Na (B) β –- and ЕС-decays of 40К (C) β –-decay of 60Co and (D) β –-decay of 137Cs. 
From practical use of radionuclides, it is clear that the most dangerous nuclides with T 1/2–30 years are 137Cs and 90Sr.įigure 17.1. So, nuclides with a long lifetime have a small initial activity, and although they emit a long time, for human life, they are less dangerous than nuclides with intermediate half-lives. But short-lived nuclides with a lifetime of the order of tens of minutes and of several hours now attract a lot of interest and are increasingly used in medical diagnostics. Nuclides with a short lifetime (<1 s) are very difficult to use, and they do not have time to bring significant harm. The military industry uses it to produce armor sheets and armor-piercing subcaliber projectiles. Having a high density (19.1 g/cm³), it is used as counterweights in airplanes and missiles in various versions of radiation protection. Depleted uranium ( 235U content is 0.2%–0.4%, in natural mixture 0.71%), formed as waste when 235U is separated from the natural mixture, is a radioactive isotope 238U, and it has a wide range of applications. For example, metal uranium can be picked up by hand, kept on a desk, drugs of uranium and thorium salts are widely used in chemistry and are in open sale. Radionuclides with a very long lifetime have insignificant specific activity. The half-life is one of the most important characteristics that determine the possibility of operating with nuclides and the danger that can be expected from them. Ilya Obodovskiy, in Radiation, 2019 17.2 Physical Characteristics of Radionuclides 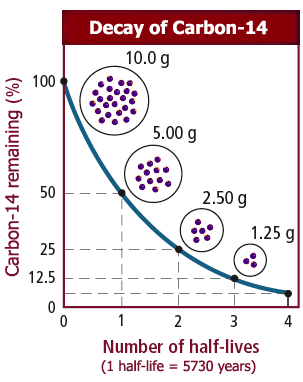
The ratio between the measured partial half-life of a particular transition and that calculated using the one-particle model for an even-even nuclei at the energy of α decay is known as the hindrance factor ( hf). release of the pre-formed α particle) in the nucleus depends on the probability of the particle hitting the Coulomb barrier times the probability of penetrating the barrier. In this model, the probability of decay (i.e. The half-life of this type of decay can be theoretically calculated using the so-called “one-body” model of α particles, which assumes that the α particle is pre-formed (as one body) within the nucleus before penetrating the Coulomb potential barrier by the tunneling effect. Since the total angular momentum for even–even nuclide is zero, and their parity is even, the ground state to ground state (gs–gs) α transitions are 0 + → 0 + transitions. The α decay of such a nuclide is a transition to another even–even nuclide. It is obvious that the lowest half-life, hence highest rate of decay, is achieved in even-even nuclides. Some have claimed that there are wide fluctuations in levels during normal labor (e.g., Cramer et al., 1971) but this has not been confirmed in other studies ( Gillard et al., 1973). There is no change following the infusion of amino acids or ingestion of a protein meal ( Tyson et al., 1971). Posture has no effect ( Ylikorkala et al., 1973), nor does strenuous physical exercise ( Lindberg and Nilsson, 1973a Pavlou et al., 1973). The factors responsible for the variation are unknown. Thus a single sample taken from an individual will not be representative of all samples taken from the individual, and the diagnostic significance of hPL levels is increased if serial levels are examined. However, the variation over a 24-hour period is greater than that which can be attributed to the assay itself ( Pavlou et al., 1972). 
Maternal levels of hPL show no nyctohemeral rhythm ( Beck et al., 1965 Samaan et al., 1966 Pavlou et al., 1972 Lindberg and Nilsson, 1973a). This is much greater than that of any other protein hormone, an interesting observation in the light of the dispute as to whether hPL has any function. The rate of synthesis of hPL has been estimated at 1–12 g/day ( Beck et al., 1965 Kaplan et al., 1968). Each subject has her own characteristic half-life and this can be attributed to differences in the mode of delivery and the physiological state of the patient. The differences in half-lives between individual subjects cannot be explained by observational error alone ( Pavlou et al., 1972). 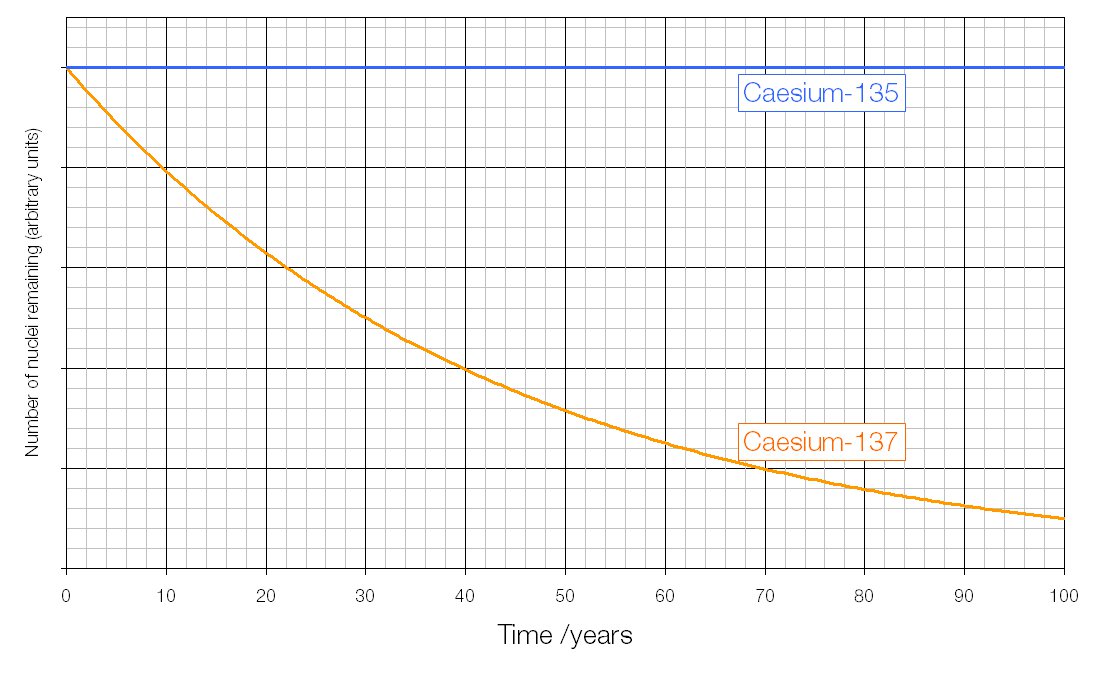
As with other biological molecules the disappearance is biphasic with an initial rapid fall (yielding the half-life stated above) followed by a period of less rapid decrease. The half-life of hPL in the mother, as estimated by the rate of disappearance after delivery of the placenta, is 10–20 minutes (Spellacy et al., 1966 Pavlou et al., 1972). Chard, in Current Topics in Experimental Endocrinology, 1983 X Metabolism and Clearance 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
